[最も人気のある!] explain the difference between heterogeneous and homogeneous mixtures 245648-What's the difference between heterogeneous and homogeneous mixtures
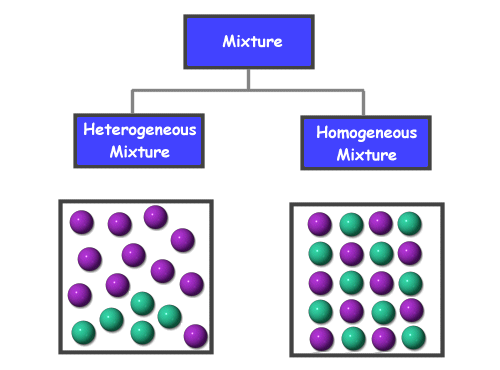
What is the difference between homogeneous and heterogeneous mixtures Get the answers you need, now!Are the following mixtures homogeneous or heterogeneous and why a vinegar b yogurt with strawberry preserves c KoolAid d acid rain e A bowl containing Skittles and M&Ms;A homogeneous mixture has a uniform composition and appearance Individual substances that constitute a homogeneous mixture cannot be visually differentiated On the other hand, a heterogeneous mixture comprises two or more substances that can be distinctly observed, and even separated relatively easily
Types Of Catalysis
What's the difference between heterogeneous and homogeneous mixtures
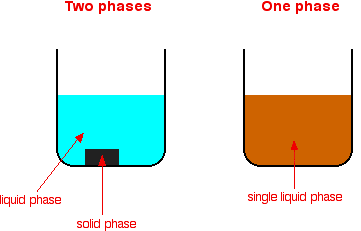
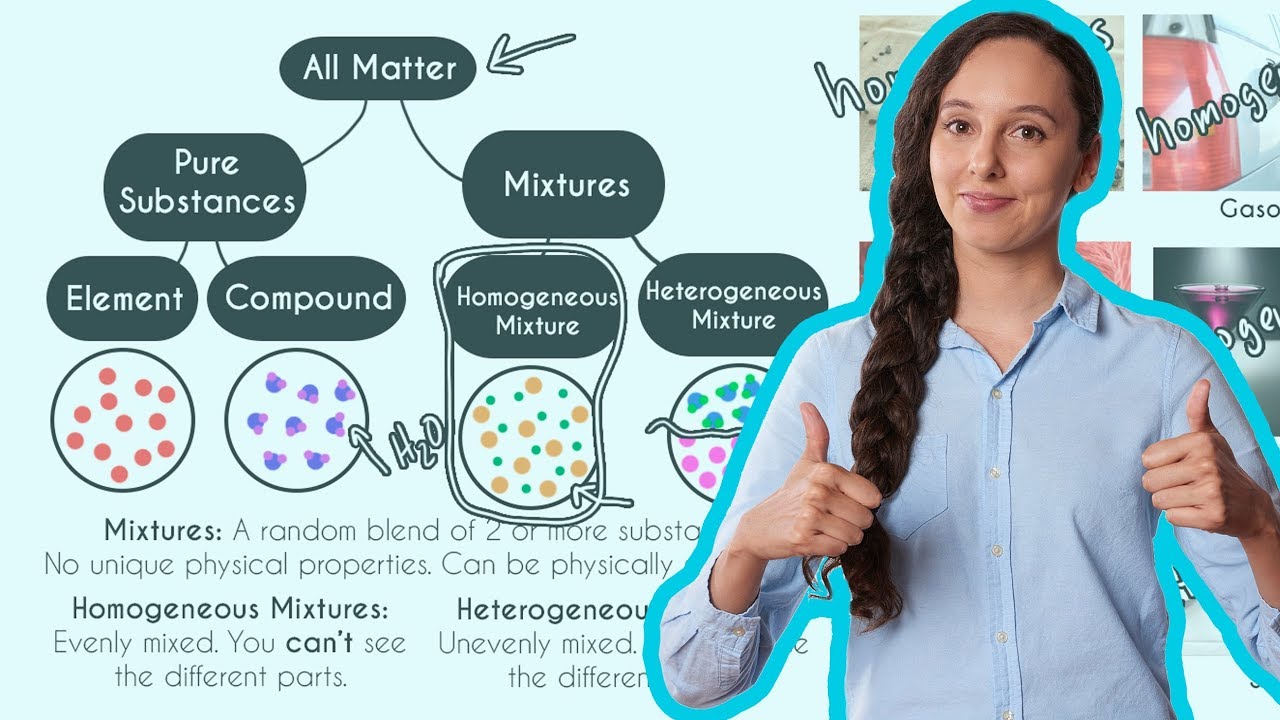
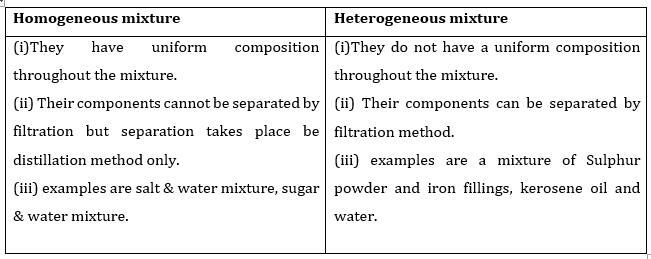
What's the difference between heterogeneous and homogeneous mixtures-Heterogeneous mixture (I) Homogeneous mixtures have uniform composition throughout the mixture (II) The whole mixture is in same phase (III) Components are not visible to the naked eye (IV) Components cannot be separated easily Eg Sugar Water → Sugar solution (I) Heterogeneous mixture have composition which may vary from point to pointThe prefixes "hetero" indicate difference A homogeneous mixture has the same uniform appearance and composition throughout Many homogeneous mixtures are commonly referred to as solutions A heterogeneous mixture consists of visibly different substances or phases



Chemistry For Kids Chemical Mixtures
Question 1 What is the difference between a homogeneous mixture and a heterogeneous mixture?More solvent can be dissolved 7 please explain me the difference between homogeneous and heterogeneous mixture with few examples And why temperature is directly proportional to rate of
Vegetable soup 5 The substance is not uniform in appearance and each part of the mixture contains a combination of different ingredients in different ratios heterogeneous mixtures 6 If a solution is saturated, which of these is true?Explain 2 Are the following mixtures homogeneous or heterogeneous TEKS describe heterogeneous and homogeneous mixtures6D explain the similarities and differences between heterogeneous and homogenous mixtures
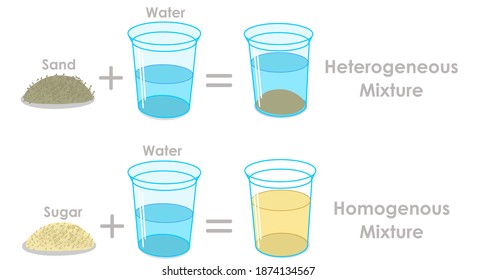
A blend of two or more kinds of matter homogeneous mixtures aka solutions, have a uniform composition;The differences between Heterogeneous and Homogeneous mixturesYou can edit this template on Creately's Visual Workspace to get started quickly Adapt it to suit your needs by changing text and adding colors, icons, and other design elements Easily export it in PNG, SVG, PDF, or JPEG image formats for presentations, publishing, and printouts The difference between homogeneous mixtures and heterogeneous mixtures is a matter of scale the heterogeneous mixture can be seen on beaches where sand included many particles like coral, shells and organic matter, etc they all can be separated easily hence known as a heterogeneous mixture but when we take a large amount of sand, it's impossible to separate all the matter, which terns as a homogeneous mixture




Difference Between Homogeneous Mixture And Heterogeneous Mixture




Examples Of Heterogeneous Mixtures Types Made Simple
The main difference between the two mixtures is that in the homogeneous you can't tell the ingredients apart whilst in the heterogeneous you can The world of chemistry and mixtures is more complex than the simple difference between homogeneous and heterogeneous However, knowing the basics is keyThis chemistry video tutorial explains the difference between homogeneous and heterogeneous mixtures within the subtopic of the classification of matter ItExplain the difference between a pure substance and a homogeneous mixture Extensive properties depend on the amount of matter that is present volume, mass, the amount of energy in the substance Intensive properties do not depend of the amount of matter that is presesnt melting point, boiling point, density tempurature, the ability to conduct electricity and heat




10 Examples Of Mixtures




Teks Describe Heterogeneous And Homogeneous Mixtures 6d Explain The Similarities And Differences Between Heterogeneous And Homogenous Mixtures 6e Ppt Download
43 The Big Bang Theory does not try to explain how the universe was created What does it explain? Difference Between Pure Substance and Homogeneous Mixture • Pure substance is made up of one component, whereas homogeneous mixture is made up of one or more components • Pure substance cannot be separated into two or more substances by any mechanical or physical method In contrast, • PureAll matter contains only one phase heterogeneous mixtures mixtures are not




Homogeneous Mixture



Lesson Categories Of Chemicals And Mixtures
The difference between heterogeneous and homogeneous mixtures is the degree at which the materials are mixed together and the uniformity of their composition A homogeneous mixture is a mixture where the components that make up the mixture are uniformly distributed throughout the mixtureHeterogeneous mixtures are those which don't have uniform properties throughout Eg colloidal substances like smoke,cloud,blood,milk Homogeneous mixtures are those which have uniform properties throughout Egbrass (CuZn) and air Homogeneous and heterogeneous are two types of mixtures that are related to the subject science, specifically chemistry The prefixes of both words help us understand the difference between the two mixtures The prefix homo means single The prefix hetero means more than one or many things




Heterogeneous Vs Homogeneous Mixtures
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures
In this animated lecture, I will teach you the concept of mixture, different types of mixture, homogeneous mixture, heterogeneous mixture, difference between The terms 'homogeneous' and 'heterogeneous' are commonly used words in chemistry and refer to solutions and mixtures Any type of mixture can be divided as homogenous or heterogeneous Because of the similarity of the terms, both are often confusing for many people just understanding the differencesSushreesamiksh53 sushreesamiksh53 Chemistry Primary School Explain the term Mixture



3




Types Of Catalysis
The terms homo and hetero indicate the most prominent differences between homogeneous and heterogeneous mixtures The prefix refers to homo while homo indicates homosexual nonhomogeneity Homogeneous mixtures have the same composition throughout the system, and heterogeneous mixtures have the oppositeHomogeneous mixture Heterogeneous mixture 1) These are called as solutions These are called as suspensions/colloids 2) Substances are Uniformly distributed These substances are Unevenly distributed 3) These are not visible to the naked eye, but visible through the microscopeWhile heterogeneous mixtures are uneven, with a composition that varies from one point to another In homogeneous mixtures, there seems to be only one component (solute and solvent), but in heterogeneous, we easily visualize more than two components




Homogeneous Mixture Examples Chemistry



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples
4 Which one of the following is not a homogeneous mixture? Refer to the explanation Homogeneous mixtures are solutions The components that make them up are evenly distributed and the appearance of the solution is uniform throughout Solutions can be solid, liquid, and gas Brass is a metal alloy (solution) composed of the metals copper and zinc Copper sulfate solution is an example of a liquid solution Air is a solution of Homogeneous mixtures are uniform, that is, their composition is the same wherever you look at it;




Homogenous Vs Homogeneous What S The Difference Writing Explained




What Is A Heterogenous Mixture Get A Clear Definition And Examples Heterogeneous Mixture Definitions Chemistry Classroom
The key difference between homogeneous and heterogeneous is that homogeneous materials and mixtures have the same uniform composition and properties throughout whereas heterogeneous materials and mixtures do not have either uniform composition or Homogeneous Mixture Heterogeneous mixture They have uniform compositions They have nonuniform compositions The components of homogeneous mixtures are not physically distinct A heterogeneous mixture has physically distinct components They have no visible boundaries of separation between the constituents6 rows Mixtures can be classified as either heterogeneous or homogeneous Homogeneous mixtures




Question Video Characteristics Of Heterogeneous Mixtures Nagwa



Lesson Categories Of Chemicals And Mixtures
41 Explain the difference between homogenous and heterogeneous mixtures 42 What is the difference between atomic number of an element and its mass number? A homogeneous mixture has only one phase throughout the solution while a heterogeneous mixture has more than one distinct phases in the mixture A phase, at least in terms of mixture, is a region in the mixture where the composition is constant wherever in that region In a homogeneous mixture (may also be called a solution), the whole of the mixture has same Heterogeneous and Homogeneous Mixtures In the lesson, you learned about heterogeneous mixtures and homogeneous mixtures Another name for a



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples



Homogeneous And Heterogeneous Mixtures Youtube
What is difference between homogeneous mixture and heterogeneous mixture?Learn from an expert tutorThis list of 10 examples of mixtures provides an explanation of whether the mixture is heterogeneous or homogeneous



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Heterogeneous Mixture Lesson For Kids Definition Examples Video Lesson Transcript Study Com
Homogeneous Mixtures Heterogeneous Mixtures centrifugation coagulation distillation evaporation filtration hand picking magnetic separation sieving winnowing sedimentation These solutions typically differ in the manner in which the solute can be separated from the solvent SolutionBased MixturesHomogenous is an older scientific term that describes similar tissues or organs It has been replaced by homologous Homogeneous is an adjective that describes similar or uniform characteristics In summary, Homogeneous is still used in modern writing Homogenous has largely been replaced by homologous Contents hide The mixing of two or more substances that results in uniform properties is called a homogeneous mixture Heterogeneous Mixture The mixing of two or more than two substances that do not result in uniform properties is called a heterogeneous mixture One can see the different particles added in a heterogeneous mixture Number of Phases Homogeneous Mixture




Homogenous And Heterogenous Mixtures Chemistry For Non Majors




Homogeneous Mixture Examples In Kitchen
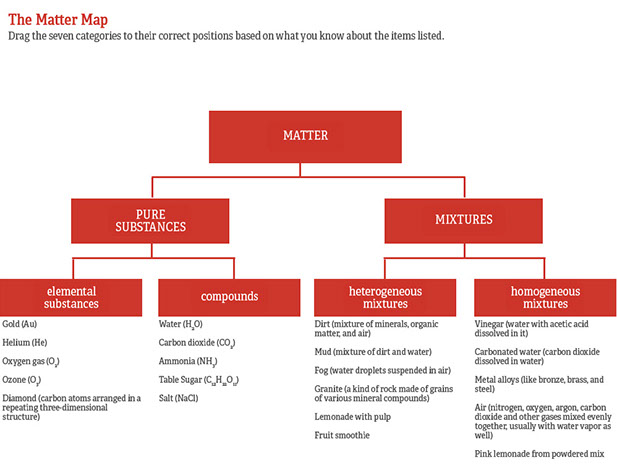
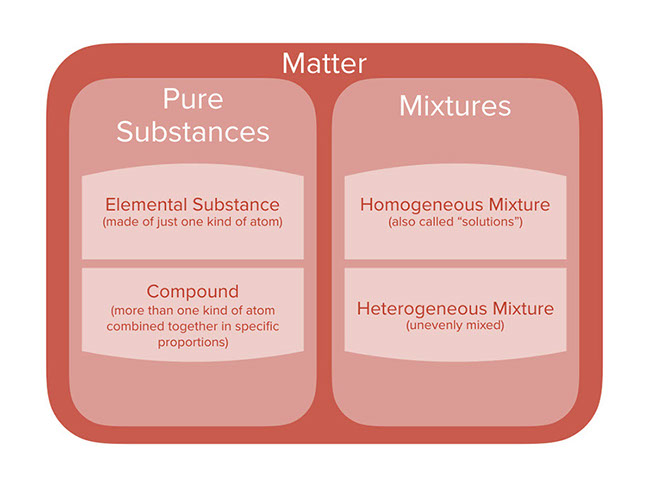
Difference Between Pure Substance and Homogeneous Mixture Pure Substance vs Homogeneous Mixture Matter is composed of different substances, like atoms and other molecules, that have volume and mass All physical objects are made up of chemical substances which are unchanging in chemical composition and characteristics A chemical substance is alsoTo summarise, Earlier, homogenous was used as a scientific term, mainly in biology, but now it is almost obsolete, being replaced by homologous Whereas, homogeneous is a common word, very much in use and means having similar or comparable characteristics Rate this article (9 / 56 votes) Email Print The Difference Between Heterogeneous and Homogeneous Mixtures Homogeneous Mixture Examples You can't pick out components of a homogeneous mixture or use simple mechanical means to Heterogeneous Mixture Examples Heterogeneous mixtures are more common than homogeneous mixtures Usually,




Homogeneous Mixture Examples Found At Home




Mixtures And Solutions Cpd Rsc Education
1 Exercise 2 Objective Type Questions Q13) Explain the term mixture Differentiate between a homogeneous and a heterogeneous mixture with one example of each in the (a) solidsolid (b) solidliquid (c) liquidliquid state of the mixture Looking to do well in your science exam ?Homogeneous and heterogeneous are words to describe mixtures Mixtures are how the particles (compounds, atoms ions bulk components) are mixed together It is the degree of44 Why do isotopes of elements have the same atomic number but different mass numbers?



Difference Between Homogeneous And Heterogeneous Welding



3
Explain the term Mixture Differentiate between Homogeneous and Heterogeneous Mixtures with one example each Get the answers you need, now!By definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers Each of the layers is Mixtures Heterogenous And Homogenous The term mixture is applied to two or more substances combined together, creating a compound combination where the different parts of the mixture have their own separate chemical identity Heterogeneous mixtures are mixtures which are made up of different substances mixed together In a mixture, the chemical bonds that exist between the different




Homogeneous Mixture Examples Chemistry




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube
Homogeneous mixtures exist in one phase of matter at a time You will not see liquid water and solid water together in a homogeneous mixture That means your glass of ice water, with ice cubes floating in it, is a heterogeneous mixture of homogeneous mixtures Homogeneous mixtures cannot be expressed as chemical formulas




Compound Vs Mixture Difference And Comparison Diffen




Homogeneous Heterogeneous Mixture Definition Examples Selftution




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii



Betrained In Cbse 9 Science Is Matter Around Us Pure Solution




Do Now Today We Will Be Discussing The Differences Between Heterogeneous And Homogeneous Mixtures Copy The Following The Words Heterogeneous And Homogeneous Ppt Download




What Is Difference Between Heterogeneous And Homogeneous Brainly In




What Is A Heterogeneous Mixture In Science Quora




What Is A Homogeneous Mixture Definition And Examples




Ch2 Questions Answers




Chemistry For Kids Chemical Mixtures



Is Matter Around Us Pure Solutions



1




10 Examples Of Mixtures



Mixtures Hd Stock Images Shutterstock
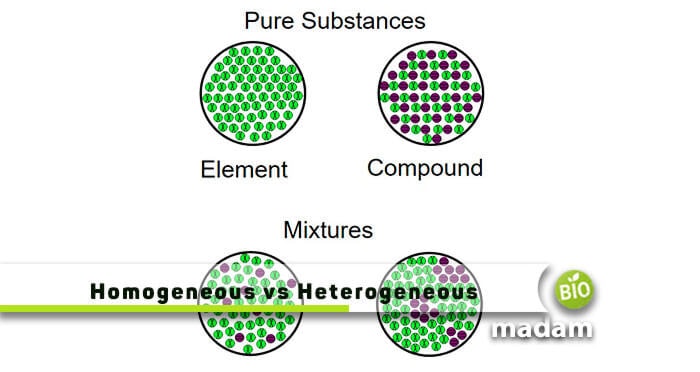



Difference Between Homogeneous Heterogeneous Mixtures Biomadam




List The Point Of Differences Between Homogeneous And Heterogeneous Mixture Brainly In




Homogeneous And Hetrogeneous Mixtures Definition Examples Teachoo



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples
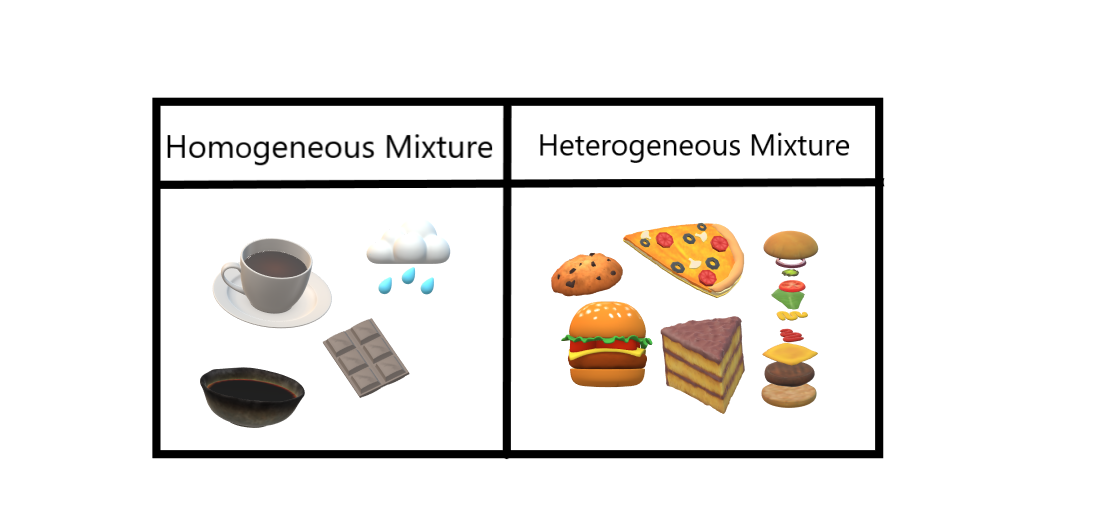



Homogeneous And Heterogeneous Mixtures Geeksforgeeks




Solutions Unit 3 Solution It Is A Homogeneous Mixture That Is Formed When A Substance Is Dissolved In Another Substance Ppt Download




What Are Some Examples Of Homogeneous Mixtures And Heterogeneous Mixtures Enotes Com




Homogeneous Mixture Definition Examples Tutors Com



Q Tbn And9gcss8oj4wp26w4gdoy1uuuzqiv502ma6xsk3cgpr6wqtj53hz7nw Usqp Cau




Difference Between Homogeneous And Heterogeneous Homogeneous Vs Heterogeneous




List The Points Of Difference Between Homogeneous And Heterogeneous Mixtures Brainly In




Homogenous And Heterogenous Mixtures Chemistry For Non Majors



Sci8u1l2



Mixture




1 Differentiate Between Homogon Eous Aid Heterogeneous Mixt Scholr



Homogeneous Mixture Examples Chemistry




Difference Between Homogenous And Heterogeneous Mixture Javatpoint




Yr4cxrz9i5bjsm




Elements Compounds Mixtures Objectives 1 Explain The Difference Between An Element And A Compound 2 Compare Heterogeneous And Homogeneous Mixtures Ppt Download




Homogeneous Mixture Examples In Daily Life



Mixture




Mixture Wikipedia




Q2 Differentiate Between Homog Lido




What S The Difference Between Heterogeneous And Homogeneous Mixtures Examples Of Mixtures Mixtures Homogeneous Mixture




Types Of Mixtures Science At Your Doorstep
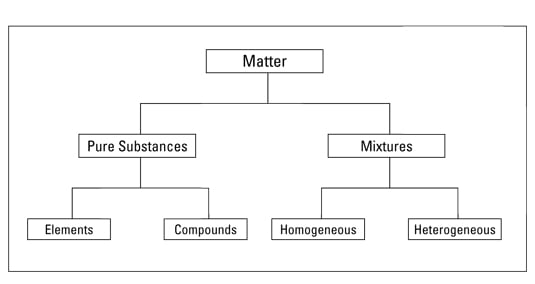



How To Distinguish Pure Substances And Mixtures Dummies



Explain I Pure Substances Vs Mixtures Uths Demo Course




What Is The Difference Between Heterogeneous Mixture Vs Homogenous Mixture Brainly Com




Mixtures And Solutions Cpd Rsc Education
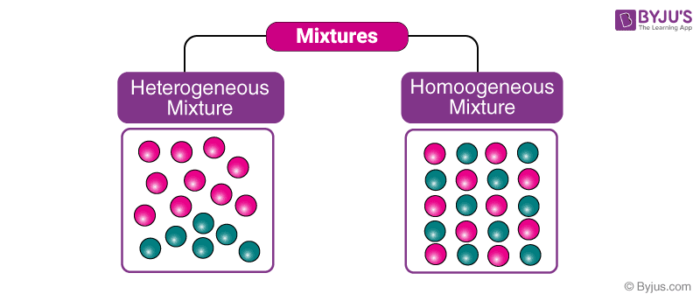



Heterogeneous And Homogeneous Mixture Differences Videos Examples



How Can Heterogeneous Catalysts Differ From Homogeneous Catalysts Quora




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Lesson Plan Homogenous And Heterogeneous Mixtures




List 5 Difference Between Homogenous Anf Heterogenous Mixture Brainly In




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Heterogeneous And Homogeneous Mixture Differences Videos Examples




3 5 Pure Substances And Mixtures Chemistry Libretexts



Http Www Nhvweb Net Nhhs Science Bklingaman Files 12 08 Ch 1 Key Pgs 16 18 Pdf




Homogeneous Mixture Definition Examples Tutors Com




Difference Between Homogeneous And Heterogeneous Compare The Difference Between Similar Terms




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Do Now Today We Will Be Discussing The Differences Between Heterogeneous And Homogeneous Mixtures Copy The Following The Words Heterogeneous And Homogeneous Ppt Download




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Difference Between Homogeneous And Heterogeneous Material Youtube




Difference Between Homogenous And Heterogenous Mixtures Youtube
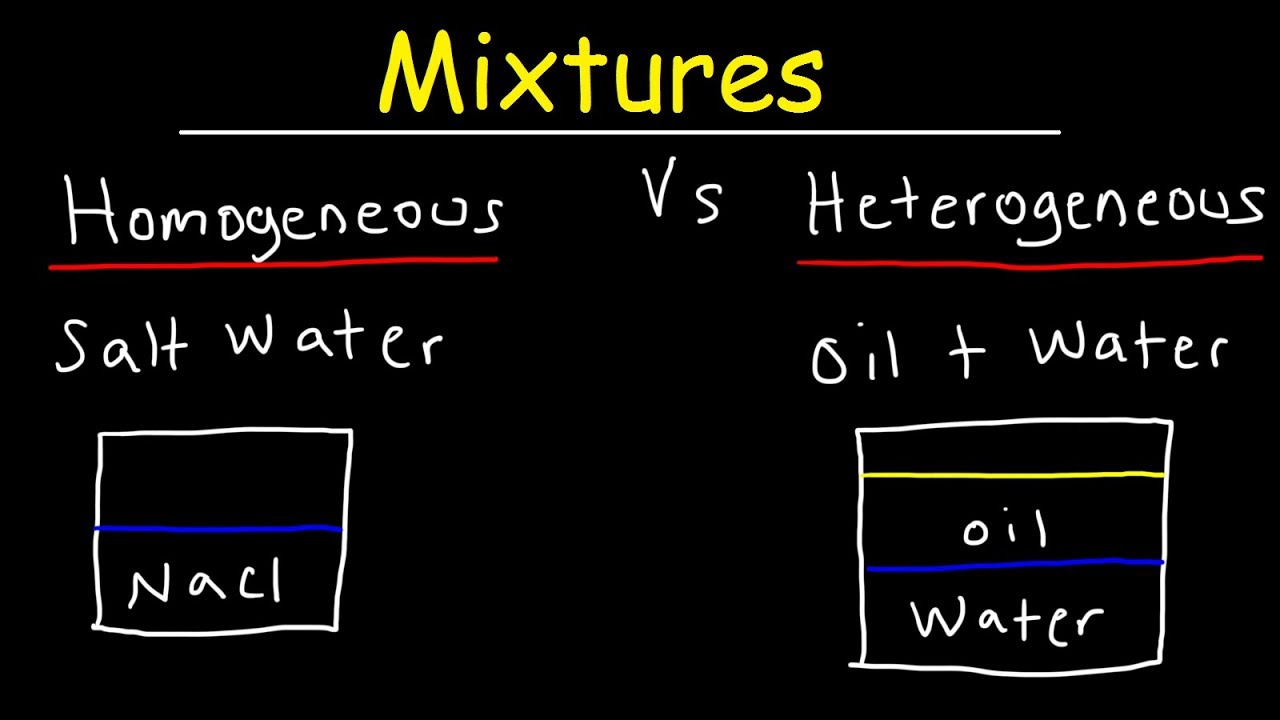



Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube
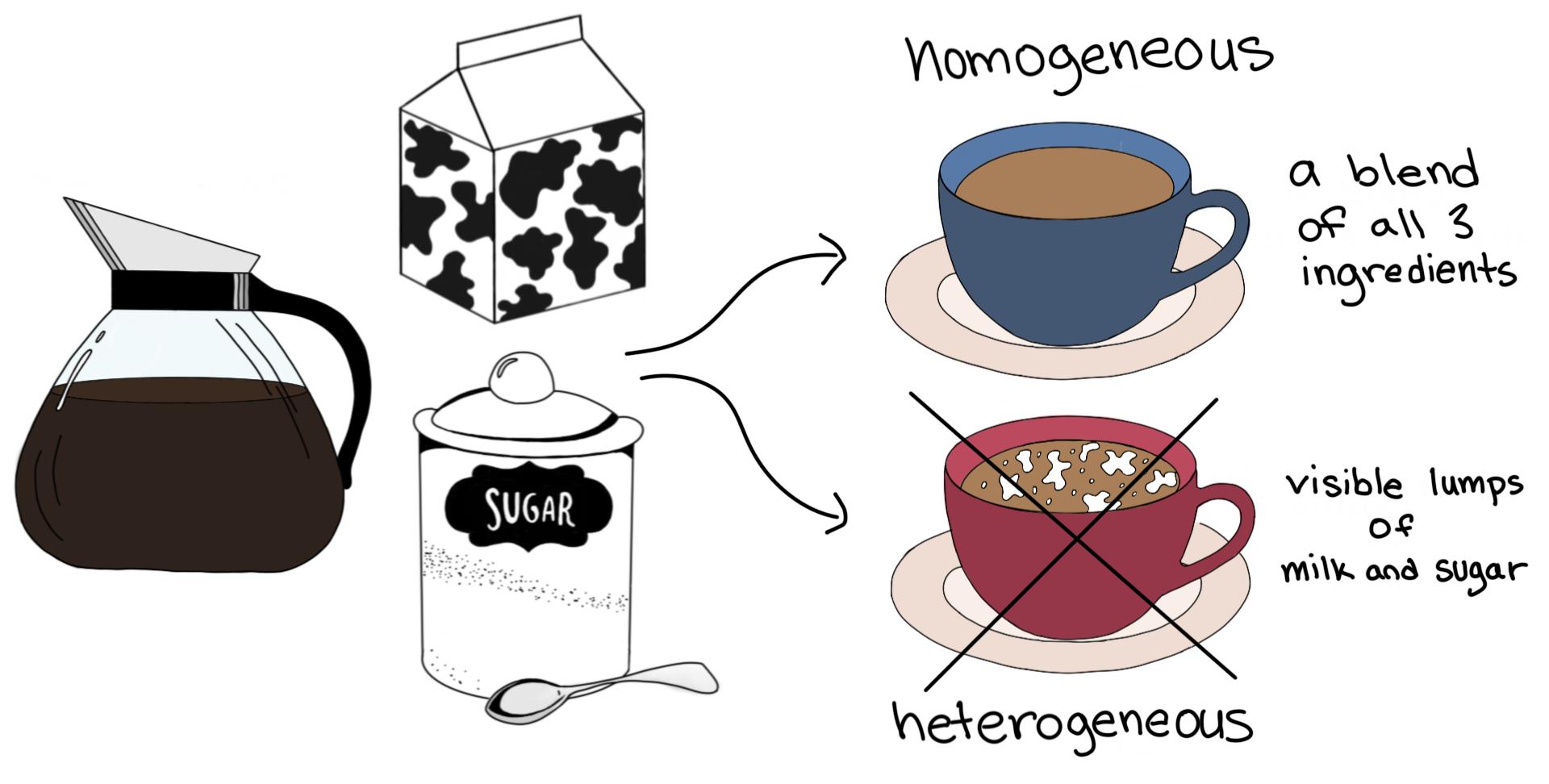



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Insprire Science Lesson 2 Mixtures And Solutions Flashcards Quizlet




Difference Between Homogeneous And Heterogeneous Mixtures Homogeneous Vs Heterogeneous Youtube
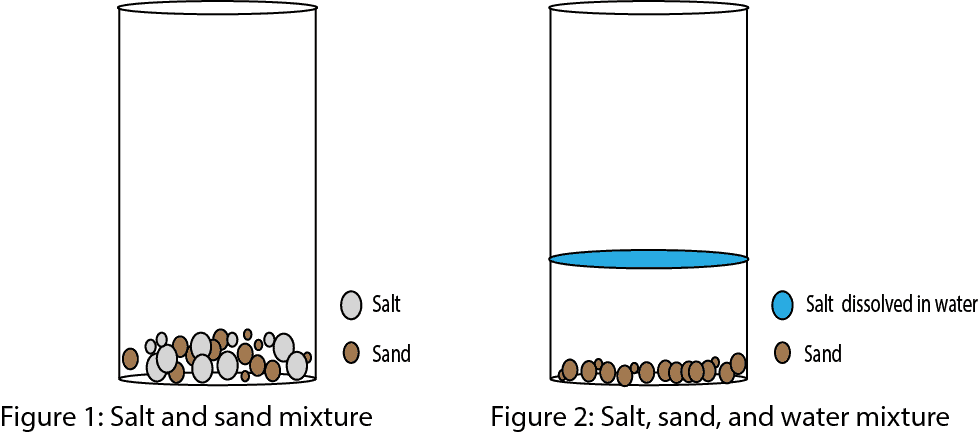



What S A Mixture How Does Heterogeneous Differ From Homogeneous Mixture And How Can Mixtures Be Separated




Heterogeneous And Homogeneous Mixtures What S The Difference Homogeneous Mixture Heterogeneous Mixture Chemistry




What Is The Difference Between A Homogeneous Mixture And A Heterogeneous Mixture Bitwise Academy




Homogeneous Mixture Definition Lesson For Kids Video Lesson Transcript Study Com



Elements Compounds And Mixtures Sas




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
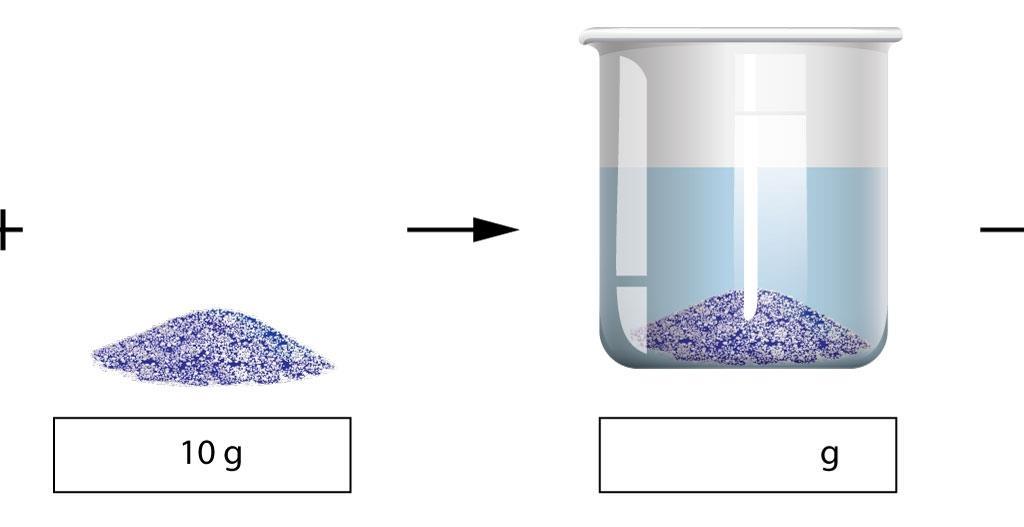



Mixtures And Solutions Cpd Rsc Education




Homogeneous And Heterogeneous Mixtures Card Sorting Activity Heterogeneous Mixture Sorting Activities Sorting Cards
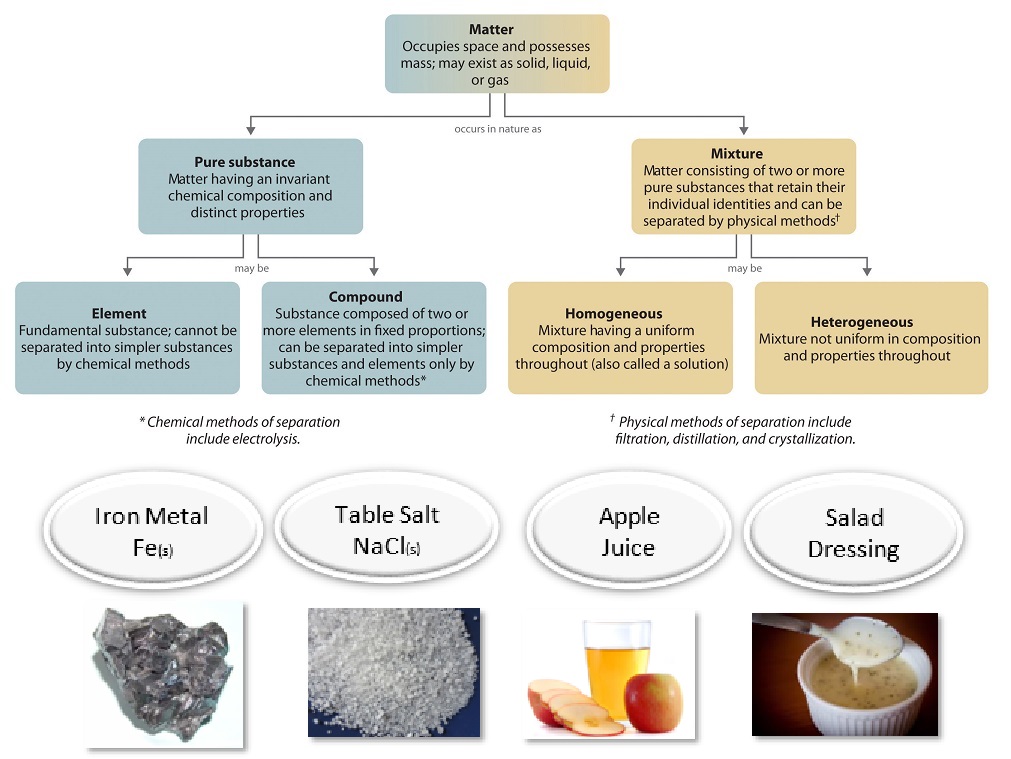



3 4 Classifying Matter According To Its Composition Chemistry Libretexts



Mixture




Homogeneous Mixture Examples In Kitchen
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures




Teks Describe Heterogeneous And Homogeneous Mixtures 6d Explain The Similarities And Differences Between Heterogeneous And Homogenous Mixtures 6e Ppt Download
コメント
コメントを投稿